Psychedelics Promote Neuroplasticity
/June 13, 2018
Spinogenesis
NEUROPLASTICITY
Mood, anxiety, and stress disorders, like major depressive disorder, generalized anxiety disorder, and Post Traumatic Stress Disorder (PTSD), are some of the most common psychological disorders. A staggering 31% of adults experience at least one anxiety disorder or PTSD1 and 21% of adults experience at least one mood disorder during their lifetime2. However, conventional medication options (like selective serotonin reuptake inhibitors [SSRIs], tricyclic antidepressants [tricyclics, TCAs], monoamine oxidase inhibitors (MAOIs), and benzodiazepines [benzos]) work slowly, taking over two weeks before benefits are experienced. Plus, about one-third of patients have no response to traditional antidepressant medications.
Degeneration of neurons in the prefrontal cortex (PFC) has been linked to depression as well as other mood and anxiety disorders. Ketamine, a dissociative anesthetic, is one of the few compounds that have been found to promote neuroplasticity in the PFC. Specifically, Ketamine has the “ability to promote the growth of dendritic spines, increase the synthesis of synaptic proteins, and strengthen synaptic responses.” Ketamine is a fast-acting and extremely effective antidepressant, even being found to work as a treatment for PTSD and heroin addiction – a “miracle” drug. However, ketamine is also known to be addictive.
Similar to the effects of Ketamine, serotonergic psychedelics (also known as classical psychedelics or serotonergic hallucinogens, like LSD) and entactogens (like MDMA) have been found to have fast-acting and long-lasting antidepressant and anxiolytic (anxiety reducing) effects with just one dose. Although not considered addictive, classical psychedelics’ therapeutic mechanisms are poorly understood due to safety concerns.
Determining a compound that could promote neuritogenesis, the development of axons and dendrites, was important because atrophy in the PFC is a contributing factor to mood and anxiety disorders. In recent studies from the University of California, Davis, cultured rodent cortical neurons were treated with a variety of psychedelics to determine what, if any, changes would occur. Most of the psychedelics produced an increase in arbor complexity, affecting both the length and total number of dendrites. Next, an 8-point dose-response study was conducted on each psychedelic and entactogen. When matched concentration-wise, most were found to be significantly more effective than ketamine at promoting neuritogenesis. Notably, LSD was found to be particularly potent. Next, in vivo effects of the psychedelics were tested on Dorsofila (a genus of flies) larvae. Early in development (first instar), both LSD and DOI promoted increased dendritic branching of class I sensory neurons, but did not affect the overall length of the dendritic arbors. When tested on more mature neurons later in development (late second instar), again, psychedelics were found to increased dendritic branching.
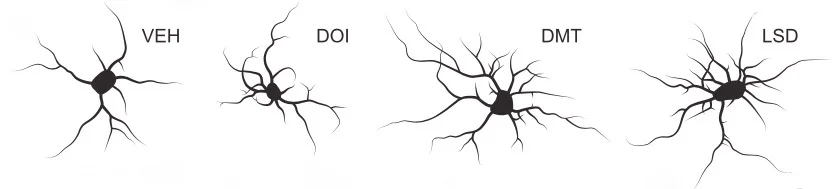
Loss of dendritic spines is also associated with depression and other neuropsychiatric disorders, so the affects of psychedelics on spinogenesis were tested. Mature rat cortical cultures were treated with LSD, DOI, and DMT (as representatives of their respective psychedelic classes, ergoline, amphetamine, and tryptamine). All three were found to increase the number of dendritic spines and promote synaptogenesis. Following, a single intraperitoneal injection of DMT into live rats was found to increase the density of dendritic spines in cortical pyramidal neurons as well as increase the frequency and amplitude of spontaneous excitatory postsynaptic currents (EPSCs), which makes the firing of action potentials more likely. These structural and functional changes were visible 24 hours later, even though the half-life of DMT is only 15 minutes.
The 5-HT2A serotonin receptor is the primary receptor responsible for the hallucinogenic effects of LSD, DOI, and DMT. Therefore, the researchers sought to determine if this receptor also played a part in their neuroplasticity-promoting effects. Treating cultured rodent cortical neurons with a combination of a psychedelic and a selective 5-HT2A antagonist, ketanserin, which basically blocks the psychedelic’s ability to activate that receptor, was found to negate the neuritogenesis and spinogenesis effects. This result signified that stimulation of the 5-HT2A receptor was key in the mechanism for the psychedelic’s neuroplastic effects.
In an attempt to further understand the neuroplasticity-promoting mechanisms, the relationship between psychedelics and Brain-Derived Neurotropic Factor (BDNF), a nerve growth factor protein known to play a role in neuritogenesis and spinogenesis, was investigated. A combined treatment of both DOI and BDNF found no additive effects, suggesting they work through similar mechanisms. Blocking the TrkB receptor, which has a high-affinity for BDNF, with a selective agonist for the TrkB receptor, ANA-12, while also treating with psychedelics found that the neuritogenesis and spinogenesis effects were blocked. This result shows that the TrkB receptor is key in psychedelics’ ability to promote neuroplastic effects. TrkB’s signaling pathway excites the kinase mTOR, which is known to promote plasticity and synaptogenesis. Treatments with a combination of a psychedelic and an mTOR inhibitor also negated these neuroplastic effects, confirming that the activation of mTOR plays a part in the mechanism through which psychedelics affect neuroplasticity.
This study gives evidential support – to the previously hypothesized but unsubstantiated claim – that psychedelics promote both structural and functional changes to cortical neurons. In both vertebrates and invertebrates, in vitro and in vivo, psychedelics showed their potential to increase dendritic branching, the density of dendritic spines, the number of synapses, and the frequency and amplitude of EPSCs. Psychedelics cannot be prescribed outright as a treatment due to safety concerns over the hallucinogenic effects and the potential for abuse. Currently, one option for patients in dire need is psychedelic therapy, where the patient remains with the psychiatrist overnight for an extensive therapy session upon taking a serotonergic psychedelic. However, the lab at the University of California, Davis proposed that non-hallucinogenic analogs of these psychedelics could be identified, which could greatly lower the safety risk and thus be prescribed for use at home. Psychedelics from four structural classes (amphetamine, ergoline, trypramine, and iboga) were found to promote neuroplasticity, with several compounds found to be more effective than ketamine. This finding greatly expands the (previously very small) number of known compounds with this sought after neuroplasticity-promoting ability. With this knowledge, new and less addictive therapeutic medications can be developed for treating mood disorders (like depression), anxiety, and other neuropsychiatric disorders through the promotion of neuroplasticity in the PFC.
Representations of treated cortical neurons
References
1 National Institute of Mental Health. (2017). Any anxiety disorder. Retrieved from https://www.nimh.nih.gov/health/statistics/any-anxiety-disorder.shtml
2 National Institute of Mental Health. (2017). Any mood disorder. Retrieved from https://www.nimh.nih.gov/health/statistics/any-mood-disorder.shtml
Ly, C., Greb, A. C., Cameron, L. P., Wong, J. M., Barragan, E. V., Wilson, P. C., ... & Olson, D. E. (2018). Psychedelics promote structural and functional neural plasticity. Cell reports, 23(11), 3170-3182. http://doi.org/10.1016/j.celrep.2018.05.022